Dissolved Substance In A Solution
Science >> Chemistry for Kids
Chemical science for Kids
Solutions and Dissolving
What is a solution?A solution is a specific blazon of mixture where one substance is dissolved into another. A solution is the same, or uniform, throughout which makes it a homogeneous mixture . Go here to learn more near mixtures.
A solution has certain characteristics:
- It is compatible, or homogeneous, throughout the mixture
- It is stable and doesn't alter over time or settle
- The solute particles are and then small they cannot be separated past filtering
- The solute and solvent molecules cannot exist distinguished by the naked heart
- It does not besprinkle a beam of light
One example of a solution is salt h2o which is a mixture of h2o and salt. You cannot see the table salt and the salt and water will stay a solution if left alone.
Parts of a Solution
- Solute - The solute is the substance that is being dissolved by another substance. In the case in a higher place, the common salt is the solute.
- Solvent - The solvent is the substance that dissolves the other substance. In the case higher up, the water is the solvent.
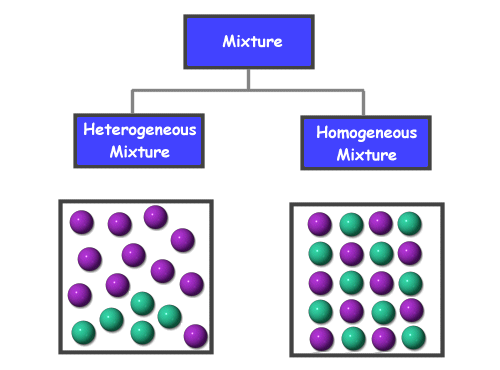
A solution is a type of homogeneous mixture
Dissolving
A solution is fabricated when one substance called the solute "dissolves" into some other substance chosen the solvent. Dissolving is when the solute breaks up from a larger crystal of molecules into much smaller groups or individual molecules. This break upward is acquired past coming into contact with the solvent.
In the case of common salt water, the h2o molecules break off salt molecules from the larger crystal lattice. They do this past pulling away the ions and so surrounding the salt molecules. Each common salt molecule still exists. It is just now surrounded by water molecules instead of fixed to a crystal of common salt.
Solubility
Solubility is a measure of how much solute can be dissolved into a liter of solvent. Call back of the example of water and salt. If you keep pouring table salt into water, at some signal the h2o isn't going to exist able to deliquesce the salt.
Saturated
When a solution reaches the point where information technology cannot dissolve any more than solute it is considered "saturated." If a saturated solution loses some solvent, then solid crystals of the solute volition start to class. This is what happens when water evaporates and salt crystals brainstorm to form.
Concentration
The concentration of a solution is the proportion of the solute to solvent. If there is a lot of solute in a solution, then information technology is "concentrated". If in that location is a depression amount of solute, so the solution is said to be "diluted."
Miscible and immiscible
When 2 liquids can be mixed to form a solution they are called "miscible." If ii liquids cannot be mixed to form a solution they are called "immiscible." An example of miscible liquids is alcohol and h2o. An instance of immiscible liquids is oil and h2o. Accept you lot always heard the saying "oil and water don't mix"? This is because they are immiscible.
Interesting Facts almost Solutions
- There is a solvent called aqua regia which can deliquesce the noble metals including gold and platinum.
- Yous can't see a axle of lite when shining it through a true solution. This ways fog is not a solution. It is a colloid.
- Solutions can be liquid, solid, or gas. An example of a solid solution is steel.
- Solids are mostly more soluble at higher temperatures.
- Carbonated beverages are made past dissolving carbon dioxide gas into liquid at high pressure.
Take a ten question quiz on this folio.
Listen to a reading of this page:
More Chemistry Subjects
Elements and the Periodic Table
Elements
Periodic Table
Science >> Chemistry for Kids
Dissolved Substance In A Solution,
Source: https://www.ducksters.com/science/chemistry/solutions_and_dissolving.php
Posted by: coxource1977.blogspot.com


0 Response to "Dissolved Substance In A Solution"
Post a Comment